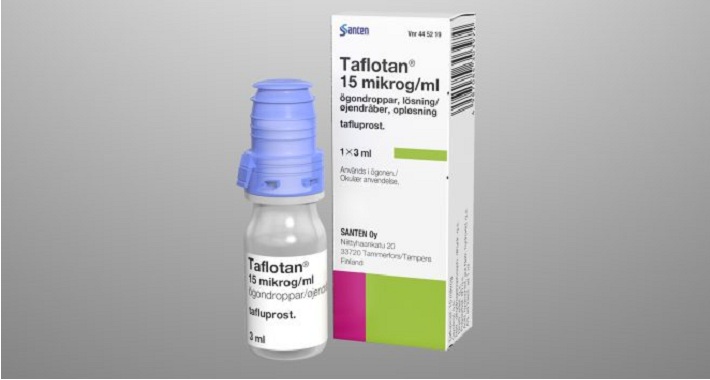
Aptar Pharma, a leading drug delivery systems provider, is pleased to announce that its innovative, preservative-free multidose Ophthalmic Squeeze Dispenser has been approved for Santen’s prescription drug Taflotan®/Saflutan® for the reduction of elevated intraocular pressure (IOP) in open angle glaucoma and ocular hypertension in 26 countries in Europe.
This approval reconfirms the market-leading position of Aptar Pharma’s Ophthalmic Squeeze Dispenser as an approved delivery system for eye disease treatment
formulations without any preservatives.
Meeting the needs of patients
Aptar Pharma is working closely with leading global ophthalmology specialist Santen Pharmaceutical Co., Ltd. to improve patient safety, achieve dosing accuracy and maintain product integrity. Santen is a specialized global pharmaceutical company dedicated to the ophthalmology field, and is spearheading the development and marketing of preservative-free drugs as part of its mission to contribute to the well-being of patients, their loved ones and society.
“We are delighted to launch preservative-free multi-dose formulations of these glaucoma therapies in Europe,” said Luis Iglesias, Corporate Officer and Head of EMEA, Santen. “By providing highly effective and well-tolerated, preservative-free medicines, with the option for multi-dose bottles, we hope to empower patients in their daily lives and improve the management of their condition.”
Chemical preservatives are often used in eye drops to ensure sterility of the drops in multidose bottles. When used in short-term treatments, they are mostly well-tolerated. For long-term use, such as in chronic conditions that include dry eye and glaucoma, preservative-free products are preferred. Preservatives have been known to cause signs and symptoms of irritation, allergy and ocular surface toxicity (1). Glaucoma patients use eye drops on a regular basis, and often for the rest of their lives.
Approval
BfArM, the German Regulatory Agency, approved Santen’s prescription drug Taflotan®/Saflutan® in the preservative-free multidose Ophthalmic Squeeze Dispenser in Germany as a first country in February 2017. Prior to this approval, Santen provided comprehensive and detailed microbiological study results as requested by BfArM. These showed that microorganisms can neither survive nor form colonies in or on the container.
The European registration process for several additional European countries ended positively and consequently national marketing authorizations have been granted based on the European harmonized evaluation. In addition, Santen performed usability studies that confirmed the user-friendliness of the multidose dispenser with glaucoma patients.
Making ophthalmic solutions easier and safer to deliver
Aptar Pharma’s Ophthalmic Squeeze Dispenser system is the result of more than 10 years of development and experience in the delivery of preservative-free ophthalmic solutions. Patients and consumers in the U.S., Europe, Latin America and Asia have benefited from this technology since 2012, with more than 175 commercial references now available on the market.(2)
“Our Ophthalmic Dispenser technology has been a great success for our customers,” explained Matthias Birkhoff, VP Business Development, Aptar Pharma. “The proven and unrivalled microbiological safety, combined with a precise and reproducible drop ejection, allows pharmaceutical customers worldwide to enter into discussions with regulatory agencies such as the German BfArM with confidence.” Mr. Birkhoff also points out the userfriendliness of the system, adding “The ergonomic, pocket-size design and the intuitiveness of a squeezable container with a low actuation force certainly contribute to the high levels of acceptance among patients and consumers worldwide.”
Staying ahead of the market
Aptar Pharma’s Ophthalmic Squeeze Dispenser provides a unique competitive advantage by addressing the worldwide trend towards patient-friendly, cost-effective and preservative-free multidose dispensers.
References:
1. EMEA Public Statement on Antimicrobial Preservatives in Ophthalmic Preparations for Human
Use. European Medicines Agency, Doc. Ref.: EMEA/622721/2009, London, 8 December 2009
2. Aptar Pharma White Paper: Available at https://pharma.aptar.com/sites/default/files/publications/aptar_pharma_osd_oct_2017_ddd.pdf
About Aptar Pharma
Aptar Pharma is part of AptarGroup, Inc., a leading global supplier of a broad range of innovative dispensing, sealing and active packaging solutions for the beauty, personal care, home care, prescription drug, consumer health care, injectables, food and beverage markets. Aptar uses insights, design, engineering and science to create innovative packaging technologies that build brand value for its customers, and, in turn, make a meaningful difference in the lives, looks, health and homes of people around the world. Aptar is headquartered in Crystal Lake, Illinois and has over 14,000 dedicated employees in 18 different countries. For more information, visit www.aptar.com/pharma
About Santen
As a specialised company dedicated to ophthalmology, Santen carries out research, development, marketing, and sales of pharmaceuticals, over-the-counter products and devices. Santen has long been the market leader in Japan for prescription ophthalmic pharmaceuticals and is now a leader in the sector in the EMEA (Europe, Middle East and Africa) region. With scientific knowledge and organisational capabilities nurtured over nearly 130 years, Santen provides products and services to contribute to the well-being of patients, their loved ones and consequently to society in more than 60 countries. For more information, please visit Santen websites www.santen.com (Japan headquarters) and www.santen.eu (EMEA).