Gene Expression Systems Explained: What Biotech Founders Need to Know Before Choosing a Platform
Key Takeaways:
- Gene expression system selection directly impacts manufacturing yield, product quality, timelines, and IP obligations
- CHO-based systems remain the industry standard for most recombinant biologics, but the specific platform within CHO matters
- GS and DHFR are the two dominant selection systems used in mammalian cell line development each with distinct trade-offs
- IP terms attached to expression platforms can create royalty and licensing obligations that persist through commercial supply
- Founders without a biology background need a plain-language understanding of these decisions before engaging a CDMO
Not every biotech founder comes from a biology background. Many of the most successful ones have come up through business development, finance, or medicine and they have learned enough science to lead a program without necessarily having deep expertise in the molecular biology underlying the manufacturing process. For those founders, certain technical decisions that their scientific team handles with apparent ease can feel opaque in ways that matter.
Gene expression system selection is one of those decisions. It happens early. It is typically led by the scientific or CMC team. And it has consequences technical, regulatory, and financial that extend far beyond the lab and well into the commercial life of the drug. A founder who understands what a gene expression system biologic drug development platform selection decision actually involves is a founder who can ask better questions, evaluate CDMO proposals more critically, and make a more informed judgment about a choice that will shape their CMC strategy for years.
What a Gene Expression System Actually Does
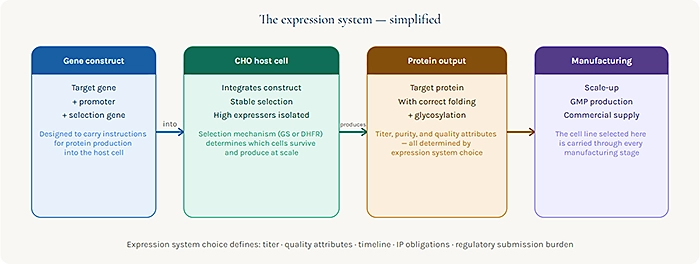
When a biologic drug a monoclonal antibody, a fusion protein, a recombinant enzyme is manufactured, it is not synthesized chemically the way a small molecule might be. It is produced biologically, by living cells that have been engineered to express the target protein according to the genetic instructions introduced by the development team.
The gene expression system is the platform that makes this possible. It encompasses the host cell line used for production, the genetic elements that control how the target gene is expressed within that cell, and the selection mechanisms used to identify and stabilize cells that express the protein at commercially useful levels. Advanced vector engineering is central to how efficiently these systems perform in practice – explore how modern vector systems are enhancing protein expression and scalability.
The choice of expression system determines how much protein the cell produces (titer), what quality attributes the protein carries (glycosylation patterns, charge variants, aggregation propensity), how stable the production cell line is over manufacturing timescales, and how well the overall system performs in a regulatory context including how familiar regulators are with the platform and how much prior characterization data exists to support the CMC package.
These are not minor variables. They define the economics of manufacturing, the complexity of the regulatory submission, and the timeline from cell line establishment to GMP material.
The Landscape of Mammalian Expression Platforms
For most recombinant biologics and particularly for monoclonal antibodies and antibody-derived formats mammalian cell expression systems are the industry standard. The reason is straightforward: mammalian cells produce proteins with the post-translational modifications, particularly glycosylation patterns, that are required for the drug to function correctly and safely in humans. Microbial systems like E. coli can produce some biologics efficiently, but for glycoproteins, mammalian systems are necessary.
Within mammalian expression, Chinese Hamster Ovary cells universally referred to as CHO cells are by far the most widely used host. Their track record in regulatory submissions is extensive, their biology is well understood, and the manufacturing ecosystem built around them media formulations, feeding strategies, bioreactor protocols is highly developed. When a regulatory reviewer sees a CHO-based manufacturing process, they are reviewing something they have seen in many forms many times before. That familiarity has real value in terms of submission efficiency.
The choice, however, does not end at “CHO cells.” Within CHO-based expression, there are several distinct platform technologies that differ meaningfully in how the expressing cell line is established and stabilized. The two most widely used in commercial biologic manufacturing are DHFR-based selection systems and GS-based selection systems.
DHFR — The Legacy Platform With a Long Track Record
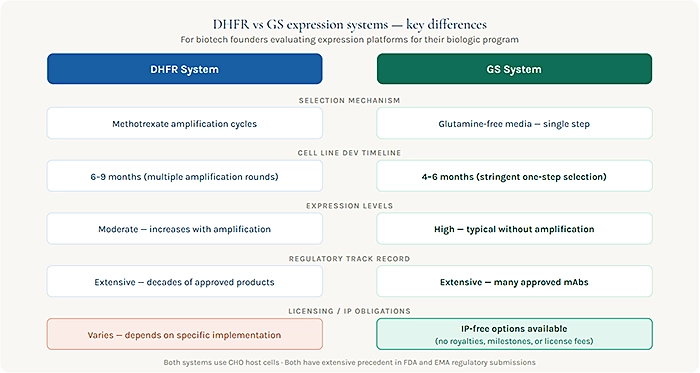
The Dihydrofolate Reductase (DHFR) selection system was among the earliest tools developed for stable CHO cell line generation and has been used in the manufacture of approved biologics for decades. In a DHFR system, the gene of interest is co-introduced with the DHFR gene into a DHFR-deficient CHO cell line. Cells that successfully integrate the construct and express the DHFR enzyme can survive in selective media conditions, and gene amplification techniques are used to increase expression levels over multiple selection rounds.
The main advantages of the DHFR system are its long regulatory history many approved products have been manufactured using DHFR-CHO systems and the substantial body of published data available to support process characterization and regulatory submissions. Its limitations include the multi-round amplification process required to achieve high expression, which adds time to cell line development, and the potential for genetic instability in highly amplified cell lines over extended manufacturing timescales.
From an IP standpoint, the DHFR system’s origins lie in academic research, and while the core biology is well in the public domain, specific implementations and cell lines carrying commercial DHFR systems may carry licensing obligations depending on the specific host and construct used. Founders should evaluate this carefully with their legal team before committing.
GS — The Modern Platform Built for Commercial Manufacturing
The Glutamine Synthetase (GS) selection system was developed specifically as a tool for commercial biologic manufacturing and has become one of the most widely adopted platforms for new biologic programs over the past two decades. In a GS system, the gene of interest is co-introduced with the GS gene into a GS-deficient CHO or NS0 cell line. Cells expressing GS can synthesize glutamine independently, allowing survival in glutamine-free selective media. This stringent selection pressure typically produces high-expressing stable cell lines without the multi-round amplification steps required by DHFR systems.
The practical advantages of GS-based cell line development are significant. Cell line development timelines are generally shorter than DHFR amplification processes. Expression levels achieved through GS selection are typically high and stable. And the regulatory track record of GS-based products which includes a substantial number of approved monoclonal antibodies means that CMC submissions built around GS cell lines are reviewed against a well-established precedent.
Lonza’s GS Gene Expression System® is the most commercially prominent implementation of GS technology in biopharma, and it is notable in the current landscape for a specific commercial reason: the company offers use of the system with no royalties, no milestone payments, and no license fees attached. For biotech founders thinking carefully about the downstream financial implications of platform selection, this matters. An expression system that carries no licensing tail means that the financial model built around the program does not need to account for royalty obligations flowing to a platform provider across the commercial life of the drug a meaningful simplification that investors and business development partners increasingly notice.
This is not the only consideration in choosing a GS-based platform, and Lonza is not the only CDMO with deep GS expertise. But it is a real-world illustration of why the commercial terms attached to an expression system deserve evaluation alongside the technical performance data.
What Founders Without Biology Backgrounds Need to Ask

For founders who are not scientists by training, engaging meaningfully with expression system selection does not require becoming an expert in molecular biology. It requires knowing the right questions to ask and being confident enough to ask them before the decision is locked.
The performance questions are the obvious ones: what titers has this system achieved for molecules similar to ours? How long does the cell line development process take from gene construct to final clone selection? What is the regulatory filing history of programs using this platform, and how have regulators responded to the CMC packages?
The commercial questions are often not asked at all, and they should be: what licensing obligations are attached to this expression system, and at what stage do they activate? Are there royalties on net sales, milestone payments at clinical or regulatory triggers, or ongoing access fees? Are these terms documented clearly in the development agreement, and have they been reviewed by legal counsel with biopharma licensing experience?
The integration questions connect the expression system choice to the broader CMC strategy: does the CDMO we are considering have an established manufacturing process built around this expression system? Will the cell line developed during preclinical work be compatible with the GMP manufacturing environment without additional engineering? Is the same organization developing the cell line also responsible for GMP manufacturing, or will there be a technology transfer between organizations?
These are not overly technical questions. They are program management and business questions that happen to live at the intersection of science and commercial strategy. A scientific team that cannot answer them clearly or a CDMO partner that is reluctant to discuss the licensing terms attached to their platform is giving you important information.
The Decision That Follows You to the Clinic
Gene expression system selection is not a decision that stays in the lab. It becomes part of the regulatory history of the program. The cell line established during preclinical development is, in most cases, the same cell line used in GMP manufacturing and carried forward into clinical trials. Changing expression systems after IND filing is technically possible but regulatory complex and almost never desirable. The decision made during preclinical CMC planning is, for all practical purposes, permanent.
That permanence cuts both ways. A well-chosen expression system one with strong performance characteristics, a clean regulatory track record, and favorable commercial terms pays dividends across the entire life of the program. It contributes to a more efficient CMC development process, a cleaner regulatory submission, and a simpler financial narrative for investors and partners.
A poorly chosen system selected under time pressure, without full consideration of its licensing implications or manufacturing compatibility creates a quiet drag that is felt in every subsequent CMC decision, every regulatory review, and every due diligence conversation.
For founders, the takeaway is simple. This decision deserves deliberate attention before it is made not a retrospective appreciation of its importance after the cell line is established and the licensing terms are signed. Bring your scientific team, your CMC lead, your legal counsel, and your financial advisor to the same table. Ask the full set of questions. And choose the platform that supports your program’s success across all of those dimensions, not just the biological ones.
Evaluating expression system options for your biologic program? Requesting a comparative technical and commercial assessment from at least two platform providers before committing is a reasonable and standard practice one that experienced CDMO partners will expect and support.




















