The CMC Bottleneck: Why Chemistry, Manufacturing & Controls is Still the #1 Reason Drug Programs miss their IND Date
Key Takeaways:
- CMC is the single most common cause of IND filing delays in biologic drug programs
- Most CMC delays are not caused by unexpected scientific failures they are caused by underestimated timelines and late starts
- Cell line development, process development, and analytical method development each carry timeline risks that compound when not planned concurrently
- Early CDMO engagement and integrated manufacturing partnerships significantly reduce CMC timeline risk
- Regulatory CMC packages require process understanding that must be built over time it cannot be compressed at the last minute
There is a particular kind of project management conversation that happens in biopharma programs with uncomfortable regularity. It usually begins with a program review six to nine months before a planned IND filing date. The clinical team is ready, the regulatory strategy is defined, the preclinical package is in order. Then someone asks about CMC status.
The CMC lead explains that cell line development took longer than projected. That process development surfaced an aggregation issue that required additional optimization cycles. That the analytical methods are still being developed and the stability studies needed for the IND package are not complete. That the GMP manufacturing slot that was reserved is now at risk because the process is not ready.
The IND date slips. The clinical timeline shifts. The board is informed. Investors ask questions.
This scenario is not exceptional. It is routine. Industry surveys consistently identify CMC delays as the leading cause of IND filing misses in biologic drug programs ahead of preclinical data gaps, regulatory strategy issues, and financing constraints combined. Understanding why CMC bottleneck IND filing drug development timeline problems persist, despite their predictability, is essential for any biotech founder or program manager who is serious about hitting a clinical milestone on schedule.
Why CMC is Persistently Underestimated
The first and most important thing to understand about CMC delays is that they are not usually caused by unexpected scientific failures. The process chemistry does not spontaneously fail. The cell line does not stop working. In the vast majority of cases, CMC delays are caused by something far more mundane and far more preventable: underestimated timelines and late starts.
The reason this happens consistently is structural. In most early-stage biotech programs, the organizational focus in the first eighteen to thirty-six months is overwhelmingly on biology on generating the preclinical proof-of-concept data that defines the program and supports the funding story. CMC is understood to be important, but it is understood as a future concern. Something to sort out once the biology is mature enough to commit to a development candidate.
This sequencing feels logical but carries a hidden cost. CMC activities cell line development, upstream and downstream process development, analytical method development, formulation development, and GMP manufacturing cannot be efficiently compressed once they start. Each stage has minimum timeline requirements driven by the biology itself. Cell line selection requires screening hundreds or thousands of clones over multiple rounds, which takes months regardless of how many resources are applied. Stability studies for the IND package require real elapsed time you cannot accelerate twenty-six weeks of accelerated stability data. GMP manufacturing campaigns require advanced booking at a facility that has the required equipment and quality infrastructure.
When these activities are started late because the program was waiting for biological certainty before committing to CMC investment the timeline math simply does not work. Every week the CMC start is delayed is a week added to the IND date, with no mechanism for recovery.
The Three CMC Workstreams That Compound Each Other
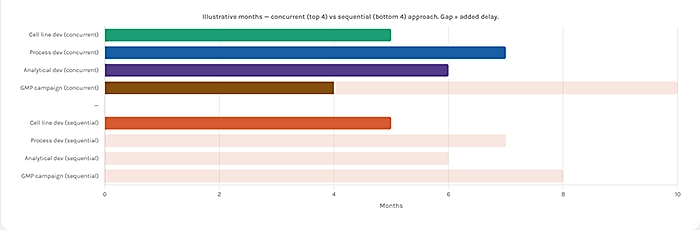
Understanding which CMC workstreams are most likely to create timeline risk requires looking at how they interact because the most damaging delays are not those that occur within a single workstream, but those that occur at the interfaces between workstreams.
Cell line development and its downstream dependencies. Cell line development is the foundation of the entire biologic manufacturing process, and it is frequently the first CMC activity to reveal timeline vulnerability. The goal is to identify a stable, high-expressing cell line that will serve as the manufacturing host for GMP production. The challenge is that this process from gene construct design through final clone selection typically requires four to six months under optimal conditions, and “optimal conditions” is not a guarantee.
Expression levels that are acceptable for research purposes may not be sufficient for commercial manufacturing economics. Post-translational modification profiles that look clean in transient expression may shift in stable cell lines. These issues are not always predictable, and when they occur, they require additional development cycles that push every downstream activity.
Critically, cell line selection is also the decision point for expression system choice. Programs that select a well-characterized, platform-compatible expression system early and ideally one with a strong track record in regulatory submissions reduce the likelihood of surprises at this stage. Systems with an established manufacturing and regulatory history reduce the analytical validation burden associated with characterizing a novel expression host.
Process development and the scale-up gap. Upstream and downstream process development is where many CMC programs discover that what worked at bench scale does not automatically translate to manufacturing scale. Bioreactor dynamics change. Purification column performance shifts. Product quality attributes that were controlled at one scale drift at another.
These are known phenomena, and experienced process development teams account for them. The problem arises when process development is compressed into a timeline that does not allow for the iterative optimization cycles that scale-up reliably requires. When a program enters process development with six months before the planned GMP campaign, and process development surfaces a significant yield or purity challenge, those six months may not be sufficient to resolve the issue, complete the required runs for process characterization, and generate the process performance data needed for the IND CMC package.
Analytical development and the stability study cliff. Of all the CMC workstreams, analytical development is the one most likely to be underestimated by program managers without deep CMC backgrounds. Developing, qualifying, and validating the assay methods required to characterize the drug substance and drug product for an IND package is a significant body of work. It requires scientific expertise, specialized equipment, reference standards, and time.
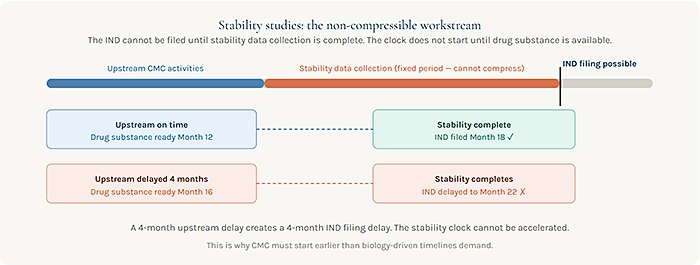
The stability study component is particularly unforgiving. ICH-compliant stability data at accelerated and long-term conditions requires real time. For an IND package, a minimum of three to six months of stability data under appropriate storage conditions is typically expected. That data generation period cannot begin until the formulated drug substance or drug product is available, which means it is entirely dependent on the completion of upstream manufacturing activities. Any delay in upstream activities pushes the stability data completion date forward, with no option for compression.
What Programs that Hit their IND Dates do Differently
The programs that consistently file their INDs on schedule are not running fundamentally different science from the programs that miss. They are managing the CMC workstream differently and the differences are visible early in the program.
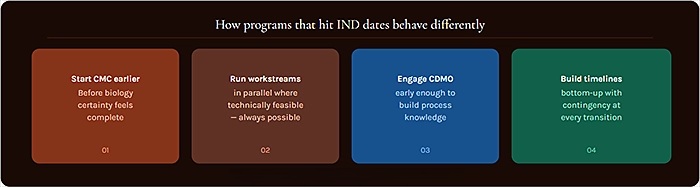
They start CMC earlier than feels necessary. The most common regret among program managers who have managed through a CMC delay is that they did not start CMC activities sooner. The biology was not quite ready, the development candidate was not quite locked, the CDMO negotiation was taking longer than expected there is always a reason to wait a few more months. Programs that hit their IND dates treat those reasons with appropriate skepticism and start CMC activities as soon as the candidate is sufficiently defined to support the work.
They run CMC workstreams in parallel where possible. Cell line development, early formulation screening, and analytical method development can proceed in parallel during the early CMC phase they do not need to be sequenced. Programs that create a detailed CMC timeline at the outset and identify opportunities for parallel workstreams consistently compress their overall timelines without compromising the quality of the work.
They engage CDMO partners early and specifically. A CDMO that is familiar with the molecule, the intended manufacturing process, and the regulatory strategy is better positioned to execute the GMP campaign efficiently and to contribute meaningfully to the CMC regulatory package. Programs that treat CDMO selection as a late-stage activity finalizing partner selection six months before the GMP campaign lose the benefit of that institutional familiarity. CDMOs like Lonza, with integrated development and manufacturing platforms, can provide continuity from cell line development through GMP supply when engaged early enough in the program to build that process knowledge.
They build realistic CMC timelines with contingency built in. CMC timelines without contingency are not plans they are best-case scenarios. Experienced CMC program managers build timelines that include buffer time at each major transition point, with explicit contingency protocols for the most commonly encountered delays. When the inevitable complication arises and it will the program has room to absorb it without the IND date moving.
The Program Leadership Responsibility
The reason CMC delays are so persistent is not that they are technically unavoidable they are largely preventable with the right planning and the right organizational commitment to treating CMC as a strategic priority from day one. The reason they persist is that they require program leadership to invest resources in CMC activities before the scientific case for investment feels fully established, and to maintain that investment even when more immediate scientific priorities compete for attention and budget.
This is a leadership challenge, not a technical one. Founders and program managers who understand that CMC is not a downstream execution function but a strategic planning function that begins at candidate selection and runs continuously through IND filing build programs that behave very differently from those where CMC is managed reactively.
The IND is not the finish line. It is the starting gate for clinical development. The programs that get through that gate on schedule are the ones where CMC was treated with urgency from the beginning not as an afterthought, but as the operational foundation on which every other program milestone depends.
Reviewing your CMC timeline ahead of an IND? The gap between where your program is today and where your CMC package needs to be for filing is worth mapping explicitly. Start with the stability studies they define the earliest possible filing date regardless of how the rest of the CMC work proceeds.




















