The US pharma tariffs policy has been formalised through an executive order signed by Donald Trump, introducing tariffs of up to 100% on imported branded medicines. The measure targets pharmaceutical companies that have not entered into drug pricing agreements with the US government or committed to establishing manufacturing operations domestically. Positioned as a national security intervention following a Section 232 investigation, the policy marks a significant escalation in the administration’s trade and industrial strategy.
Policy Framework and Tariff Structure
The executive order outlines a tiered tariff mechanism centred on compliance with pricing and onshoring requirements:
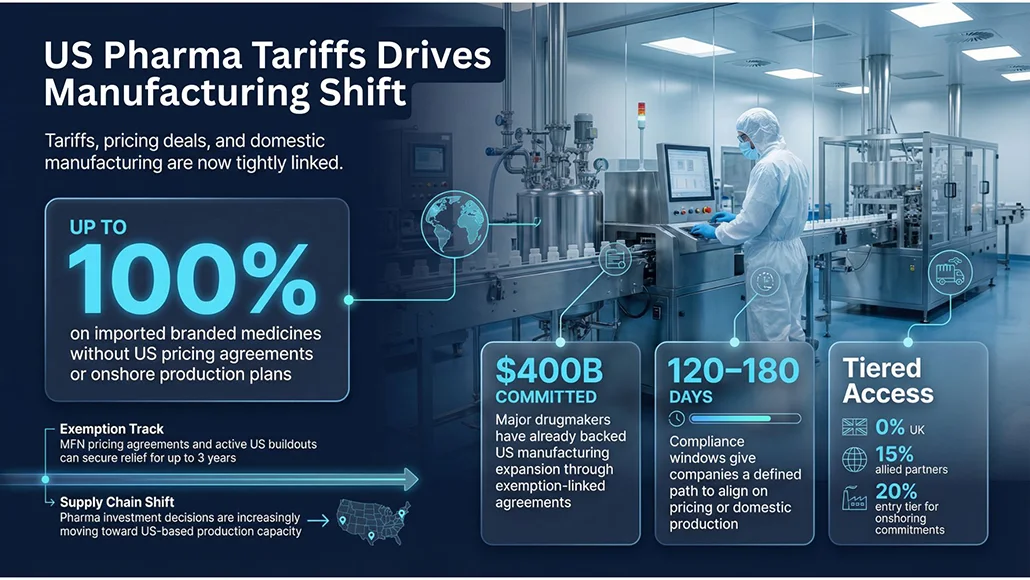
- Up to 100% tariffs on patented medicines and their active pharmaceutical ingredients (APIs) that are not produced in the US and lack pricing agreements
- 20% tariff for companies committing to establish US manufacturing, rising to 100% after four years if conditions are unmet
- 15% tariff applied to imports from trade-aligned regions including the EU, Japan, South Korea, and Switzerland
- 0% tariff window for the UK under a separate bilateral trade agreement, linked to increased domestic drug pricing commitments
- Exemptions for generic drugs, biosimilars, and certain specialty medicines meeting urgent public health needs
Companies that have signed Most Favoured Nation (MFN) pricing agreements or are actively negotiating with the US Department of Health and Human Services while building domestic capacity are exempt from tariffs for up to three years.
Compliance Timelines and Industry Participation
The administration has introduced defined compliance timelines to accelerate industry response. Larger pharmaceutical companies are given 120 days to align with pricing or manufacturing requirements before facing full tariffs, while smaller firms have 180 days. According to officials, 13 drugmakers have already finalised agreements, with negotiations ongoing with four additional companies.
Major industry players such as Pfizer, Eli Lilly, and Novo Nordisk have secured exemptions through prior agreements tied to the MFN pricing framework. These arrangements have contributed to approximately $400 billion in commitments to reshore pharmaceutical manufacturing into the US during the current administration.
Regulatory Context and Trade Policy Alignment
The US pharma tariffs policy follows a Commerce Department investigation that concluded pharmaceutical imports pose a potential threat to national security. This sector-specific approach operates outside the scope of a prior US Supreme Court ruling in February, which invalidated broader global tariffs imposed under emergency powers but did not extend to Section 232-based measures.
The announcement coincides with the anniversary of the administration’s “Liberation Day” tariff strategy, reinforcing a broader shift toward protectionist trade policies and domestic industrial reinforcement. In parallel, the administration has also revised tariff calculations for imported materials such as steel, aluminium, and copper, further tightening supply chain cost structures.
Operational and Market Impact on Drugmakers
The policy introduces significant structural implications across the pharmaceutical sector, particularly in manufacturing and supply chain strategy. While large multinational companies have largely mitigated immediate exposure through agreements, smaller and mid-sized firms face disproportionate challenges.
The Midsized Biotech Alliance of America has warned that the tariffs risk creating a two-tier system, favouring companies with diversified portfolios and capital capacity. Smaller innovators, often reliant on contract manufacturing and limited product pipelines, may struggle to absorb increased costs or rapidly shift production to the US.
Industry data indicates that around half of drugmakers represented by PhRMA have not yet secured agreements, leaving a substantial portion of the market exposed to tariff escalation. Legal challenges have already been initiated by industry groups contesting the framework.
Strategic Implications for Global Pharma Supply Chains
The introduction of US pharma tariffs is expected to accelerate localisation strategies and reshape global pharmaceutical supply chains. Companies are increasingly evaluating capital allocation toward US-based manufacturing infrastructure to secure tariff exemptions and maintain market access.
At the same time, differentiated tariff treatment for allied trade partners signals a recalibration of international pharmaceutical trade flows. Countries with existing agreements retain partial access, while others may face reduced competitiveness in the US market.
From an industry perspective, World Pharma Today observes that the policy reinforces a dual objective- cost containment through pricing agreements and supply chain security through domestic production mandates. This alignment is likely to influence long-term investment decisions, regulatory engagement strategies, and cross-border manufacturing partnerships across the pharmaceutical sector.