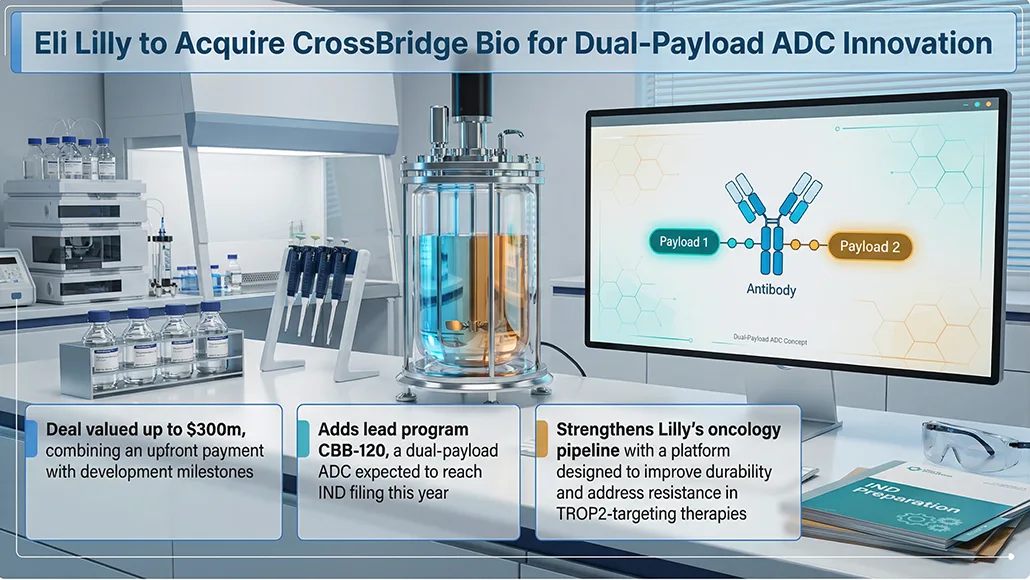
Eli Lilly and Company has entered into an agreement to acquire all outstanding shares of CrossBridge Bio in a transaction valued at up to $300m, structured through an upfront payment and additional milestone-based compensation linked to development progress. The deal centres on strengthening Lilly’s oncology pipeline, with a particular focus on advancing next-generation antibody-drug conjugates (ADCs). As Lilly acquires CrossBridge, the move signals a targeted expansion into dual-payload ADC technologies designed to address complex cancer treatment challenges.
The acquisition brings with it CrossBridge Bio’s lead candidate, CBB-120, a dual-payload ADC engineered for oncology applications. The therapy is expected to reach a US Food and Drug Administration (FDA) investigational new drug (IND) application stage within the year. Developed using a platform created by Kyoji Tsuchikama at the University of Texas Health Science Center at Houston, the technology combines two mechanisms of action within a single construct. Specifically, camptothecin-based bioconjugate-120 integrates a topoisomerase I (TOP1) inhibitor with an ataxia telangiectasia and rad3-related protein (ATR) inhibitor, targeting trophoblast cell surface antigen 2 (TROP2) to improve efficacy and durability in treatment outcomes.
Founded in 2023 and headquartered in Houston, CrossBridge Bio has positioned its platform to overcome resistance pathways commonly observed in current TROP2-targeting ADCs while enhancing the therapeutic index. The company has attracted backing from investors including Alexandria Venture Investments, CE-Ventures, Linden Lake Labs, Portal Innovations, and Texas Medical Center Venture Fund, alongside support from the Cancer Prevention and Research Institute of Texas. Advisory support for the transaction includes Cooley as legal counsel and Zwick Advisory providing strategic guidance to the board.
CrossBridge Bio co-founder and CEO Dr Michael Torres said: “This transaction will allow Lilly to advance our new generation of dual-payload antibody-drug conjugates, including CBB-120, with the potential to meaningfully improve outcomes for patients with limited treatment options.
“At CrossBridge Bio, we believe our dual-payload ADC platform is uniquely positioned to be transformative in oncology. I’m proud of how well our team has executed and advanced our platform in such a short time since the company’s founding.
“By becoming a part of Lilly, a leader in patient-focused therapeutic development, we are well-positioned to further accelerate the clinical potential of this approach.”
As Lilly acquires CrossBridge, the integration of early-stage assets spanning preclinical to Phase I development underscores a strategic bet on differentiated ADC innovation within oncology.