Introduction
The clinical research industry wants to achieve the common goal of accelerating drug development by improving the process of data collection, data validation, analysis, and submission. Faster time to market puts pressure on every aspect of trial including Clinical Data Management (CDM), arising the need for effective, efficient and near real time data management.
Generation of good quality data and patient safety are basic principles of good clinical practice. All groups including CDM should work together to achieve it with patient centric approach. Cross functional Agile teams should work collaboratively to enhance health outcome and quality of life.
Agile practices can be applied as solution at all phases of CDM i.e., setup, conduct and closeout. Traditional CDM process follows waterfall method and is sequential in nature. This may lead to non-utilization of resources and increased wait time, which impacts overall study setup turnaround time (TAT) and peripheral activities such as documentation, analysis, and submission. Lack of agility around people, process, technology, and workplace would lead to following operational challenges.
People: The process of document creation, review, and approval leading to delayed feedback and increased waiting time. Multiple functions working together create key dependencies and less collaboration impacts the quality and TAT of deliverables.
Process: One final database release (eCRFs and all Edit checks) before first patient In. Lack of ownership due to command-and-control way of working. Longer TAT for creating specification, User Acceptance Testing (UAT) etc. and no global library.
Technology (Tools and Technique): Extensive use of spreadsheets for tracking and reporting. Standard estimation techniques based on assumptions. Manual scheduling and tracking of projects.
Workplace: Traditional setup and closed functional group.
Understanding the road blockers for adoption of Agile framework is critical.
Agile framework for CDM
Adaptation of Agile framework can help mitigate challenges of traditional CDM process efficiently. Agile values and principles can be adopted by leveraging various framework, for example- Crystle, eXtreme Programming, Scrum, CBO Agile practices etc. The most relevant frameworks for CDM are:
- Scrum provides an iterative, incremental approach to enhance predictability and control risk. It involves groups of people who collectively have all the skills and expertise to do the work and share or acquire such skills as needed. This is suitable to CDM study in which the involvement of different functional team members like Data Manager, Programmer etc. is essential and collaborative work is needed. This can bring synergy in cross functional teams and streamline the process by reducing waste and focusing on the critical, leading to accomplishment of customer’s goal.
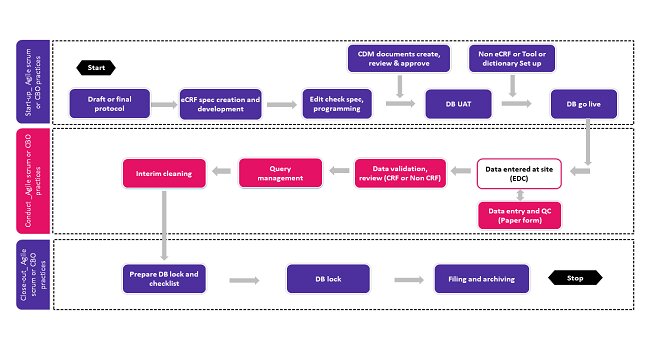
- CBO Agile Practices provide light weight approach for activities that are ongoing and transactional in nature. CBO Agile Practices like collective wisdom, collective work management, visualization of work and embrace change can be utilized as per requirements without major modification in process.Figure: CDM landscape and Agile feasibility check
Figure: CDM landscape and Agile feasibility check
Transformation with Agile framework
People: Empowering, self-organized, cross functional small team will be more successful as compared to hierarchical, controlled, and functional large team.
Process: Instead of large release, go for small releases (like Go-Live with eCRFs and EDC functionality checks or standard edit checks or critical edit checks etc.). Continuous feedback and iterative process will deliver quicker than linear process.
Tools and Technique: Automation, continuous release will help speed up process and delivery.
Workplace: Collaborative culture and synergy between roles will deliver value to customer.
a. Scrum framework in CDM
Scrum provides framework for Agile implementation in CDM. Instead of extensive planning, plan adequate scope for few weeks (sprint planning). In daily connects, discuss the progress, and plan your action for next 24 hours (daily Scrum), review outcome, work on next move (sprint review), inspect entire sprint cycle (sprint retrospective) and start new sprint with identified improvements in earlier sprint along with sprint backlogs.
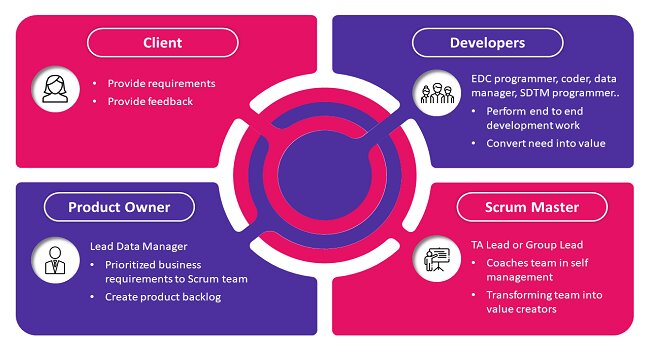
Figure: Mapping of Agile Scrum roles in CDM team (product owner, Scrum master and developers)
Implementation of Scrum in CDM
- In Study setup, batches of eCRF and edit checks will be delivered through Sprint.
- Based on the stages of the studies, Agile Scrum framework will be implemented in conduct or closeout-
i. Scrum milestone studies: Studies that are already nearing a milestone
ii. Scrum instream data cleaning: Studies with no milestone but have subjects who have completed study participation or defined visits.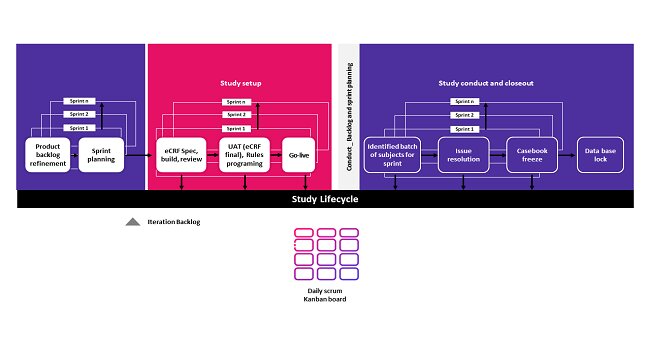
Figure: Scrum implementation in CDM
b. CBO Agile Practices
In Scrum, each sprint length cannot be more than defined time limit. However, in CDM, the sprint length may vary based on various factors such as the phase of the study, study design, therapeutic area, and any other study complexity drivers etc.
Complete Scrum framework implementation may have certain pre-requisite like tailoring of SOPs or WI creation and aligning team to Agile Scrum operating model. Scrum perspective dashboard, metrics and KPI need to be defined and agreed. Also, change management is a critical parameter to embark on Agile method of CDM delivery.
CBO Agile practices like collective work management, visualization of work, collective wisdom and embrace change will ensure service reliability. Value log, Scrum team and sprints collectively enhance value of Scrum, leading to customer satisfaction. Implementation of these practices is feasible without major changes in process.
In Business 4.0 era, CBO Agile practices can be implemented in CDM, which will significantly impact the delivery time, enhance efficient teamwork, and make workflow easy.
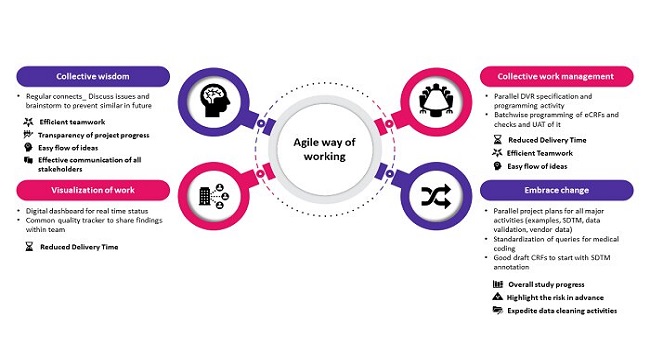
Figure: Agile way of working in CDM
Agile CDM Framework’s value add
Implementing Agile Framework in CDM has the following benefits:
- Breaking siloes and collective thinking
- Daily Scrum will reduce multiple to-and-fro email communication within the team and across
- Early validation and risk mitigation
- Delivering quality data output
- Affluence to end users
- Building parallel programming and database to avoid rework
- Customer engagement right from the beginning
- Real time good documentation practice
- Clinical database can be modified and change at any stage of the project
- Transparency, as every stakeholder knows the progress
- Early predictability of study progress
Quantitative benefits
- Up to 40% reduction in final protocol approval to go live and LSLV to DB lock timeline.
- Up to 50% reduced timeline of round table UAT.
- Improved TAT of CRF development and edit check build by 20-40% by using sprint and better control with sprint metrics.
Qualitative benefits
- Real time issue management.
- More accountable, empowered, and independent teams lead to reduced oversight and handoffs.
Conclusion
Transformed role, independent and flexible process and technology driven methods will be future landscape of CDM. Change is inevitable and readiness to adopt it is the only way forward. CDM process transformation with Agile approach will help to accommodate this change requirement. Application of Agile approach can shift clinical data management from traditional siloed process to agile way. Every team needs to pick required methods i.e., Agile Framework- Scrum or CBO Agile practices to align with internal and external customer objective and to achieve the goal of better performance. Agile way of working demands flexibility that needs to respond quickly to customer needs and by changing direction as per the situation demands. Key of successful implementation will be through robust training, right skillsets, collaborative work environment, empowered people, and team. Agile methodology is a shift in mindset to achieve the goal and business driven process emphasizing the business value over process.
References
- https://www.scrum.org/resources/scrum-guide
- https://agilemanifesto.org/
- Scrum Narrative and PSM Exam Guide; 2021 Edition, Author-Mohammed Musthafa Soukath Ali
- https://knome.ultimatix.net/wikis/2213-one-click-agile
Author – Shivaji Bote is a Subject Matter Expert for CDM Domain in TCS life sciences.
Co-Authors – Late Akash Nilawar was the Agile Ninja Coach for CDM Domain in TCS life sciences.
Sharad Sharma is a domain consultant in TCS Life Sciences.
Dr. Jayathirtha Gopalakrishn comes with two-decade of Pharmaceutical/ Clinical Research experience.