Imagine the Possibilities: What if innovations in the life sciences supply chain kept up with innovations in medicines and treatments?
With many serialization and track & trace regulations in the compliance period, and others (e.g., EU FMD, US DSCSA) on the horizon, life sciences companies need to think beyond serialization as a core capability and prepare for the next generation of supply chain. Verifying the provenance of a medicine—from where its ingredients were made and where the drug was manufactured, to how the medicine was handled through the end-to-end supply chain and all the way to the patient in a trusted manner, is becoming an increasingly mandatory supply chain capability.
That scenario is now possible using blockchain technology.
Blockchain (sometimes referred to as Distributed Ledger Technology, or DLT) is protected by cryptography, that allows a network of nodes to collectively maintain a shared ledger of information without the need for complete trust between the nodes. Blockchain is essentially a time-sequenced chain of events based on an agreed upon consensus mechanism. The mechanism guarantees that, as long as the majority of the network validates the entries (i.e., the “blocks”) posted to the ledger (i.e., the “chain) as per stated governance rules, information stored on the blockchain can be trusted as reliable.
Investors and enterprises across multiple industries and functions have taken notice of blockchain’s potential. Recently, investment and spending on blockchain-based technology have each topped more than $1 billion and continues to accelerate. Accenture’s current projections for the blockchain services market alone estimates a CAGR of more than 60 percent, hitting close to $7 billion by 2021. Within life sciences alone, blockchain technology could provide a $3 billion opportunity by 2025.2
THE CASE FOR BLOCKCHAIN IN LIFE SCIENCES
While significant investment has gone into serialization capabilities over the past decade, life sciences companies are restricted by traditional technology limitations where information is captured in a database, transmitted to another database (owned by a separate internal or external partner) and then reconciled.
Blockchain is becoming increasingly relevant to address the perennial challenges of trust and speed in the life sciences supply chain. For example, enabling data driven business decisions in forecasting the flow of goods and having visibility into the origin of materials and inputs has always been difficult, partly due to the technologies involved and partly because suppliers are mistrustful about sharing data they fear will be abused by downstream customers, impacting their ability to price products effectively.
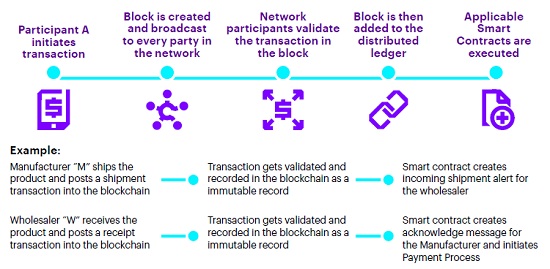
Blockchain’s ability to generate trust and create the right permissions around the sharing of data allows life sciences companies to break through some of those commercial challenges, enabling better traceability. Companies can access the information they want without revealing commercially sensitive data. For example, complications associated with the recall process are dramatically streamlined through concurrent notification within the supply chain.
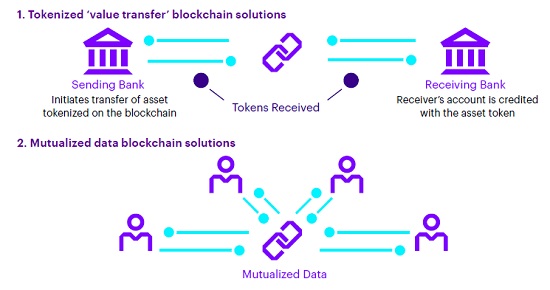
There are several types of blockchains that can drive value for the life sciences supply chain, depending on the need and types of permissions among those sharing information.
Different types of blockchains include:

One important attribute of blockchain technology is smart contracts. Smart contracts are event-driven programs which run on a distributed ledger and can take custody over an asset on that ledger. The embedded “IF, THEN” statements allow for automatic transactions based on pre-defined terms. For example, a payment can be remitted when certain criteria are achieved in a transaction on the blockchain.
PRACTICAL APPLICATIONS ACROSS THE LIFE SCIENCES SUPPLY CHAIN
Blockchain has applicability across all supply chain functions. However, it can particularly address certain unique aspects of the life sciences supply chain as there requirements of the supply chain evolve with the product and therapy mix.
BENEFITS OF BLOCKCHAIN TECHNOLOGY
Immutability/indisputability: Single source of truth, replicated across all nodes; data on the ledger is encrypted, pervasive and persistent
Automation: Network self-validates all ledger entries; smart contracts automatically enforce business rules; near real-time data and transaction processing
Cost reduction: Reduction in operational costs related to exceptions and reconciliation
Auditability: Provides a real-time track-and-trace audit trail;improves business, operational and regulatory reporting
Decentralization: Assets are tied and controlled by their owners rather than institutional custodians; exchange of information with pre-agreed consensus validation instead of third-party
Security: Encryption provides record-level security of data; no single point of failure—network is resilient against attacks on individual nodes
PROVENANCE
One of the most complex life sciences supply chain challenges has been the ability to effectively track the origin of a product (or therapy) from raw materials to the finished product. Despite the various efforts of full chain of custody systems that exist today, the fragmentation of systems between trading partners opens the risk for fraud. Blockchain technology is an ideal solution, given that no single organization is responsible for provenance. Organizations across the life sciences ecosystem benefit from having authentic product in the supply chain, ensuring brand integrity and improved patient outcomes by delivering authentic product to the patient. Blockchain enables the idea of a “digital passport” for a product, containing all relevant information for each component or ingredient, including instructions and patient adherence information from the packaging.
SERIALIZATION TRACK AND TRACE
For the last two decades, regulators across the globe have been implementing requirements for unique product identification to deliver greater security of finished products through the supply chain. These requirements have the aspiration to eliminate counterfeit and diverted products, ultimately contributing to increased patient safety.
Like provenance, one of the major challenges with track and trace is the effective exchange of data across the ecosystem of partners – from the pharmaceutical manufacturers, to wholesale distributors, to dispensers. With the use of blockchain,supply chain partners can more effectively and securely share data across the supply chain and, eventually, with the end patient.
A significant business opportunity exists in using blockchain with serialization capabilities for the recall process. Recall notification, once injected into the blockchain, can initiate communication and alert messages to all affected parties (manufacturers, distributors, dispensers and eventually patients). All parties can track and verify the recalled product, minimizing the time it takes to dispose of the recalled products, reducing risk and costs.
SPECIALTY LOGISTICS
In one aspect of the life sciences supply chain, pressure is mounting on pharmas to optimize their cold chain logistics. This series of uninterrupted temperature-controlled, refrigeration, production, storage and distribution of products is complicated and expensive. The pharmaceutical industry expects to spend nearly $17 billion by 2020on cold chain management.4
When applied to the cold chain, blockchain combined with technologies like IIoT (Industrial Internet of Things) can create secure documentation of storage temperatures at every point in a product’s journey. This enables supply chain managers and executives to identify potential temperature excursions and other efficiencies across the end-to-end supply chain, and also presents an opportunity to the challenges posed by CAR-T, as previously mentioned. Additional opportunities exist to complement blockchains with artificial intelligence (e.g., machine learning) to detect potential specialty logistics issues.
BEYOND THE SUPPLY CHAIN:LAST MILE TO THE PATIENT
Only 38 percent of patients feel knowledgeable about new products coming to market that may benefit their health and less than half of patients feel that their doctors discuss the entire spectrum of therapies, which often leads to treatment decisions being made without the ‘big picture’ being understood. Once all the product data in the supply chain is stored in the blockchain, life sciences organizations will be able to target patients with far more relevant information—such as product availability, detailed product information, and specific product history. The ability to tie the patient to the product will not only increase patient satisfaction, but ultimately their ability to manage their own health for better outcomes. Inevitably, blockchain will more effectively deliver personalized medicines to patients in the right setting at the right times, with the right dose.
OVERCOMING OBSTACLES
Compared to the financial markets (which themselves have yet to achieve blockchain maturity), the life sciences industry’s participation with the technology remains in its infancy. The following represent the key barriers the technology must overcome to gain a legitimate place in life sciences:
NETWORK EFFECT. To reap full value from blockchain, the majority of partners in the ecosystem must participate. Interoperability between blockchains and other technology solutions need to be addressed to get full ecosystem participation.
GOVERNANCE. Smart contracts require agreement on allowed transactions among the ecosystem partners and is an ongoing exercise—this requires strong ecosystem participation and buy in.
REGULATORY AND LEGAL. Because blockchain technologies offer a new socio-political paradigm for doing business, few legal and regulatory frameworks are in place to govern their use. From a life sciences perspective, strong regulatory frameworks can ensure the integration of blockchain technology to avoid challenges in terms of regulatory reporting, HIPAA compliance, etc. Regulators also need an in-depth understanding of how to operate in a blockchain environment.
PERFORMANCE. As the blockchain in data and number of nodes, the time to verify transaction may increase causing potential delay to standard business actions.
STARTING THE BLOCKCHAIN JOURNEY
Within the next three years, approximately 30 percent of life sciences companies plan to utilize blockchain (if they aren’t already), opening new business opportunities and addressing challenges of the past.5 Supply chain applications of blockchain are endless for life sciences companies given the requirements for specialized medicines and therapies. Those that start now—who are willing to experiment, fail fast and innovate based on that experience—will be the organizations to achieve competitive advantage and improve patient outcomes.
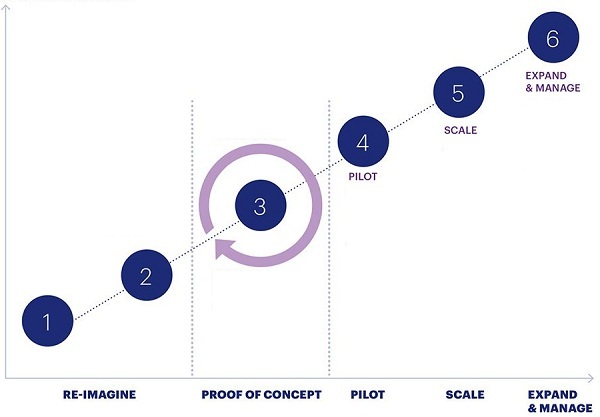
1.Strategy
Set vision for how Blockchain can support the business challenges or opportunities
2. DEVELOP & PLAN
Define the specific business areas for blockchain value Develop plan to execute PoC(s)on high value areas and opportunities
3. PROOF OF CONCEPT
Controlled learning of Blockchain Technology with a focus on select use case Incorporate learnings from PoC phase into pilot
4. PILOT
Continued learning of the Blockchain based use case Complete high-level assessment and roadmap to transition
5. SCALE
End-to-end implementation of Blockchain based solution for targeted business challenge, aligned to strategy
6. EXPAND & MANAGE
Expand platform to additional business units and deploy on going improvements
REFERENCES
1 Accenture Research
2 Accenture projection
3 Accenture Research
4 2017 Biopharma Cold Chain Sourcebook, Pharmaceutical Commerce (www.PharmaceuticalCommerce.com), Brooklyn, NY
5 Accenture Research