The Hidden Cost of Licensing in Biologic Drug Development – and Why More Biotech Startups are Rethinking it
Key Takeaways:
- Platform licensing costs in biopharma are often underestimated in early financial modelling
- Royalty stacks can compound across multiple technology licenses, creating downstream deal complexity
- IP-free or royalty-free expression systems can meaningfully simplify financial projections
- Investors and boards are increasingly scrutinizing licensing obligations in due diligence
- Evaluating licensing terms during platform selection not after is the strategic move
Most early-stage biotech founders are extremely focused on the science. That is appropriate you cannot build a program around a molecule that does not work. But there is a second category of decision that shapes the fate of a biopharma company just as profoundly, one that tends to receive far less attention in the earliest stages: the licensing obligations attached to the platforms and technologies used to develop and manufacture your drug.
These are not obscure fine-print concerns. They are financial realities that will show up in your cap table conversations, your board presentations, your CDMO negotiations, and eventually, in the deal terms of any partnership or acquisition discussion you enter. For companies developing biologics monoclonal antibodies, fusion proteins, bispecifics, and related modalities understanding the full scope of licensing costs in biologic drug development biopharma is not optional. It is foundational.
Where Licensing Costs Actually Come From
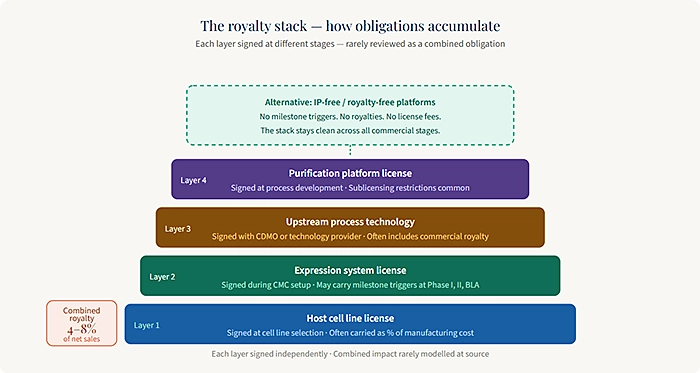
The licensing landscape in biopharma is dense, and for biologic drug programs specifically, it spans multiple layers. There are the obvious ones: platform access fees, expression system licenses, and cell line licensing agreements. Then there are the less obvious ones the downstream royalty obligations that activate when your drug enters clinical trials, achieves regulatory approval, or reaches commercial milestones.
Each of these layers is individually negotiable, but they are rarely evaluated together as a stack. A company might license a cell line from one provider, use an expression system from another, and access a manufacturing platform through a third. Each agreement carries its own royalty rate, its own milestone triggers, and its own sublicensing restrictions. When viewed in isolation, each seems manageable. When stacked together, they can represent a significant and ongoing drag on the program’s economic profile.
The issue compounds as programs advance. In pre-IND stages, licensing fees are often modest and easily absorbed. But as you move into GMP manufacturing, Phase I trials, and eventual commercial supply, the royalty rates and milestone triggers that seemed theoretical become very real obligations. By that stage, the licensing stack is already locked in. The time to evaluate it was earlier.
What Royalty Stacks Do to Financial Models
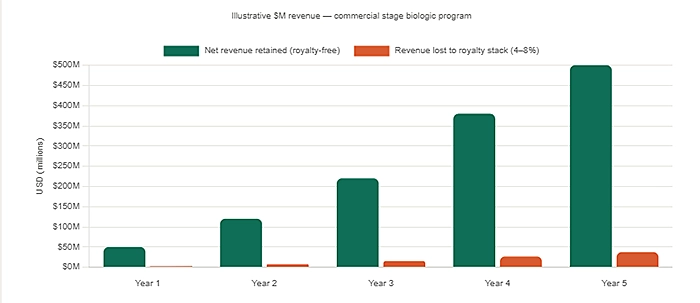
The financial modelling implications of a heavy licensing stack are worth understanding in concrete terms. When a biologic drug program carries multiple royalty obligations each percentage point drawn against net sales or manufacturing cost the downstream revenue projections change materially. For a product with peak annual sales of $500 million, even a combined royalty rate of four to five percent across multiple licenses represents tens of millions of dollars per year in obligations that flow out of the program before any return reaches investors or the company itself.
For early-stage companies, this is not a commercial-stage problem only. It is a fundraising problem right now. Institutional investors particularly those evaluating pre-Series B or Series B biopharma companies have become considerably more sophisticated in their financial due diligence. They look at licensing stacks. They model out the royalty obligations against projected revenues. They assess how those obligations affect net margins and, critically, how they affect the attractiveness of the asset in a partnership or M&A scenario.
A clean financial model, with minimal or no licensing obligations attached to core platform technologies, is simply easier to defend. It gives investors a cleaner line of sight from development spend to commercial return. It removes a layer of deal complexity that can slow down term sheet negotiations and reduce the final valuation of an acquisition. Founders who understand this dynamic will make different choices at the platform selection stage because that is where the model is built.
The Rise of IP-Free and Royalty-Free Platform Approaches
The good news is that the market has responded to this problem. Over the past decade, a meaningful number of CDMOs and platform providers have moved toward IP-free or royalty-free licensing models for their core manufacturing and expression technologies, recognizing that reducing the IP burden on their clients creates better long-term partnerships and more competitive positioning.
One well-documented example is Lonza’s GS Gene Expression System®, which the company offers with no royalties, no milestone payments, and no license fees attached. The commercial rationale is clear: by removing the downstream licensing obligations, Lonza positions itself as a development and manufacturing partner rather than a technology licensor, which creates a different kind of long-term relationship with biotech clients. Programs that use the GS System own the economics of their own success rather than sharing them with a platform provider across the commercial life of the drug.
This is not the only IP-free approach in the market, but it is a useful illustration of how platform selection decisions carry financial implications that extend years into the future. When evaluating expression systems, cell line technologies, and manufacturing platforms at any stage of development the licensing terms attached to those technologies deserve the same scrutiny as the technical performance data.
The Conversation Your Legal and Finance Teams Need to Have Early
There is a structural reason why licensing costs tend to be underweighted in early-stage decision-making: the people making platform selection decisions are often scientists or program managers, while the people who feel the downstream financial consequences are the CFO, the board, and eventually the investors. The bridge between those two groups the conversation that translates platform choice into financial model impact does not happen automatically. It needs to be designed in.
The most effective early-stage programs build a cross-functional platform evaluation process that explicitly includes legal and finance representation. Before committing to an expression system, a cell line technology, or a manufacturing platform, the team should have a clear answer to three questions. What royalties, milestones, or fees are attached to this technology, and at what triggers do they activate? How do these obligations interact with any other licensing commitments already in the program? And how do these terms affect the attractiveness of the program in a future partnership or acquisition scenario?
These are not difficult questions to ask. They are often simply not asked at the stage where they should be during platform evaluation, not during Series B due diligence.
Negotiating Better Terms When You Cannot Avoid Licensing

Not every platform technology comes with a royalty-free option, and there are situations where a proprietary technology with licensing obligations is genuinely the right scientific choice for a program. In those cases, the goal is not to avoid licensing it is to negotiate the most favorable terms possible and to document those terms clearly for future investors and partners.
A few principles are worth keeping in mind. Milestone triggers are often more negotiable than royalty rates. If you can push milestone payment triggers to later clinical stages Phase II completion rather than Phase I entry, for example you preserve more capital during the highest-burn early development period. Caps on total royalty payments over the commercial life of the product are sometimes available and worth pursuing. Sublicensing provisions that preserve your ability to partner or sell the program without triggering punitive fees should be a standard ask.
None of this is to suggest that licensing terms are always adversarial or that platform providers are not legitimate contributors to the value of your program. Many are. The GS expression system, to return to the earlier example, was developed through decades of research and represents genuine scientific value. The point is simply that the terms attached to that value whatever provider you are working with should be negotiated deliberately, with full awareness of the downstream financial implications.
Protecting the Economics of Your Own Success
There is a broader principle at work here that goes beyond any single licensing negotiation. Building a biologic drug program is expensive, time-consuming, and risky. Most programs that start do not succeed. For those that do succeed that reach Phase I, generate positive clinical data, and attract the attention of larger pharma partners the financial reward should flow to the people and organizations that took that risk: the founders, the investors, the employees.
Licensing obligations that were negotiated quietly during platform selection can meaningfully reduce that reward. Not because any single agreement is unreasonable, but because the stack of obligations, accumulated across multiple decisions made under time pressure and scientific focus, adds up to a financial burden that was never fully modelled and never fully approved by the people who bear its consequences.
Getting this right is a matter of process. It requires that licensing evaluation is built into platform selection as a formal step, not an afterthought. It requires that finance and legal are at the table during decisions that are often treated as purely scientific. And it requires that founders develop enough financial literacy around IP structures to ask the right questions before they are locked in.
The economics of your success are worth protecting. The time to start is before the first licensing agreement is signed.

Evaluating platform options for your biologic program? Make sure your expression system and manufacturing platform evaluation includes a full IP and licensing assessment alongside the technical data. The two are inseparable.




















