Key Takeaways:
- Lead selection is not just a scientific decision it carries significant CMC and regulatory consequences
- Molecule attributes like expression level, stability, and manufacturability should be factored in during lead identification
- Early CMC planning aligned with lead selection reduces costly late-stage surprises
- Platform-based manufacturing systems can reduce lead-to-IND timelines when selected early
- Biotech founders who involve CMC expertise at the lead stage protect both their timeline and investor narrative
There is a moment in early drug development somewhere between the excitement of a promising target and the sobering complexity of clinical planning when a critical decision gets made without nearly enough deliberation. That decision is lead selection. Which molecule moves forward. Which candidate becomes the program.
For most biotech teams, this choice is driven almost entirely by efficacy and potency data. That is understandable. You are building a drug, after all, and a drug needs to work. But the consequences of lead selection extend far beyond biology. The molecule you choose to advance will define your Chemistry, Manufacturing, and Controls (CMC) roadmap for years and in many cases, it will either enable or quietly strangle your path to the clinic.
This article is written for biotech founders and biopharma program managers who want to understand why lead selection CMC strategy drug development decisions are deeply interconnected, and what it costs when that connection is ignored.
The CMC Implications Nobody Talks About in Early Discovery
When a discovery team presents lead candidates for selection, the data on the table typically includes binding affinity, selectivity profiles, in vitro potency, and perhaps some early ADME results. Rarely does it include a CMC risk assessment. Rarely does anyone ask: how does this molecule behave at scale? What does its expression profile look like? How stable is it under manufacturing stress conditions?
These are not downstream questions. They are selection criteria.
A molecule that performs beautifully in a small-scale research setting can become a manufacturing nightmare at the 200-liter bioreactor scale required for Phase I material. Expression levels that look acceptable in a transient system often fall short in stable cell line development. Post-translational modifications glycosylation patterns, charge variants, aggregation tendencies that go unnoticed in early assays can trigger regulatory questions that delay your IND filing by six to twelve months.
The uncomfortable truth is that some molecules are simply harder to manufacture than others. And choosing one of those molecules, without fully appreciating the manufacturing implications, means your CMC team inherits a problem they did not create and cannot easily solve.
What Manufacturability Actually Means

Manufacturability is often treated as a vague, post-selection concern. It should not be. In practical terms, a manufacturable biologic is one that can be produced at sufficient yield, with acceptable purity, within a reproducible process that regulators can audit and approve.
For monoclonal antibodies, this often comes down to expression system selection, host cell compatibility, and how cleanly the molecule can be purified. For fusion proteins or more complex modalities, the challenges compound. Molecules with high hydrophobicity, unusual disulfide bonding patterns, or a tendency to aggregate will demand more process development time, more analytical method development, and more regulatory justification.
None of this is insurmountable. But all of it takes time, and in early-stage biotech, time is capital.
This is why the concept of developability assessment has gained traction over the past decade. Leading CDMOs and internal CMC teams now routinely run in silico and wet-lab developability screens on lead panels before final candidate selection. The goal is not to select the “easiest” molecule it is to go into selection with open eyes, understanding the trade-offs between biological performance and manufacturing complexity.
Integrating CMC Thinking into Lead Selection Practically
The gap between discovery and CMC does not have to be structural. There are practical ways to bring CMC thinking into the lead selection process without slowing science down.
Developability flags as selection inputs. Running a short-panel developability screen on your top three to five leads adds two to four weeks of data that can meaningfully inform the selection discussion. Key parameters to evaluate include thermal stability (Tm by DSF), colloidal stability (kD by DLS), expression titer in a representative cell system, and basic aggregation propensity. These data points do not override efficacy but they create a more complete picture of what you are choosing.
Expression system alignment early. The gene expression system used for lead characterization should ideally mirror what will be used in manufacturing. If your CMC pathway involves a GS (Glutamine Synthetase) system such as Lonza’s GS Gene Expression System®, which offers freedom to operate without royalty obligations then running your leads through that system during characterization gives you a meaningful, apples-to-apples comparison. Data generated in a transient HEK system may look very different from data generated in a stable GS-CHO system, and reconciling that difference post-selection adds avoidable complexity.
Cross-functional lead selection reviews. Formalizing a cross-functional review step one that includes at least a CMC representative or a CDMO partner before final candidate selection is one of the highest-leverage process changes an early-stage biotech can make. It does not require a large team. It requires that CMC is at the table before the decision is locked.
The Downstream Cost of Getting Lead Selection Wrong
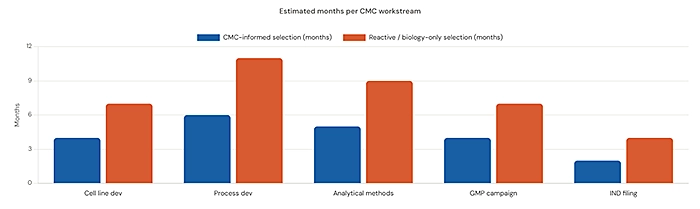
Program managers and founders need to understand what happens when a molecule with CMC challenges is selected without that knowledge being factored in. The consequences are not always visible immediately they often surface six to eighteen months later, exactly when the program needs to be accelerating.
Process development timelines extend. A molecule that requires additional engineering to improve expression or reduce aggregation can add three to six months to your CMC timeline before you even begin GMP manufacturing runs. If you are on a standard IND timeline, that time does not exist.
Analytical complexity grows. Challenging molecules require more robust analytical characterization. Each additional assay is additional development time, additional validation, and additional regulatory documentation. This is not just a science problem it is a resource problem.
Regulatory risk increases. The FDA expects CMC packages to demonstrate process control and product understanding. A molecule with complex critical quality attributes and a difficult manufacturing process requires more supporting data to demonstrate that control. If that data is incomplete or inconsistent, reviewers issue information requests that delay your IND filing.
Investor narrative suffers. For biotech founders navigating pre-Series B or Series B fundraising, CMC risk is increasingly on investors’ radars. Clean financial models, tight CMC timelines, and well-characterized development candidates are significantly easier to defend in a data room. A program carrying unresolved CMC uncertainty is a harder story to tell.
What a CMC-Informed Lead Selection Process Looks Like
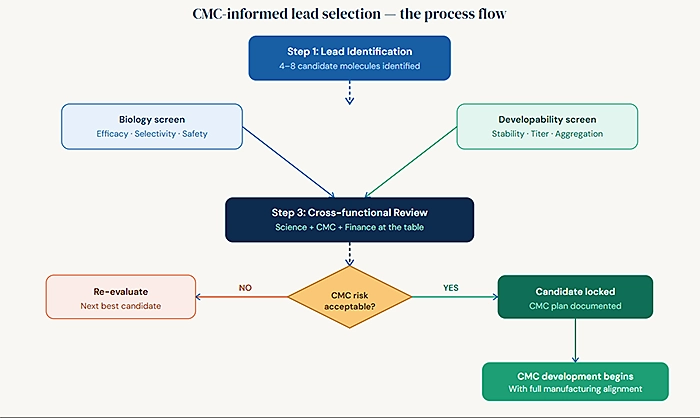
The best-run early-stage programs treat lead selection CMC strategy drug development as a unified exercise rather than two separate activities happening in sequence. The process typically looks like this:
During lead identification, a small set of molecules usually four to eight candidates are evaluated in parallel using a standardized developability screen. This screen is designed not to predict manufacturing performance exactly, but to surface any outliers that carry unusual CMC risk.
When the data comes back, the selection team reviews it alongside efficacy and selectivity data. The default selection criteria remain biology-first. But if two candidates show equivalent biological performance and one has materially better developability characteristics, that information is used. The team also uses this stage to begin preliminary discussions about manufacturing platform because the choice of expression system, cell line, and CDMO partner should be informed by what the molecule can support.
After selection, the CMC team has a fully documented rationale for why this candidate was chosen, which includes a transparent acknowledgment of any known manufacturability considerations and how the CMC development plan addresses them. This documentation becomes part of your regulatory file and your due diligence package.
The Competitive Advantage of Getting It Right
There is a version of drug development where CMC is treated as a support function something that follows discovery and exists to execute the plan that scientists hand over. That version produces programs that are slower, more expensive, and more prone to avoidable delays.
The programs that consistently reach the clinic on time and arrive there with investor confidence intact are the ones where CMC is treated as a strategic function from the beginning. Where lead selection is understood as the foundation of the CMC plan, not a separate decision made before the CMC plan begins.
For founders, this means expanding what you look for in a development candidate. Efficacy will always matter most. But the molecule that gets you from IND to Phase I fastest is not always the most potent one. Sometimes it is the most manufacturable one.
That distinction is worth understanding before you make the most consequential selection decision in your program’s history.

Interested in how your lead selection process aligns with your CMC roadmap? Evaluating expression system choices before candidate lock is one of the highest-leverage decisions early-stage programs can make. Start the conversation with your CMC team or CDMO partner earlier than feels necessary you will not regret it.




















