Schreiner MediPharm, a Germany-based global provider of innovative functional label solutions for the healthcare industry, is supplying its durable Pharma-Comb IL Label solution to a company whose clinical trial cancer medication requires low-temperature storage and pre-administration thawing, making marking and documentation particularly challenging.
Various clinical trials are currently focused on investigating the systematic use of oncolytic viruses in cancer therapies. Shanghai Yuan Song Biotechnology is among the companies engaged in this research. For its trial relating to innovative virotherapy, Schreiner MediPharm supplies the appropriate specialty label, complete with various integrated functions, that easily withstands temperatures of -20 till -70 degrees Celsius.
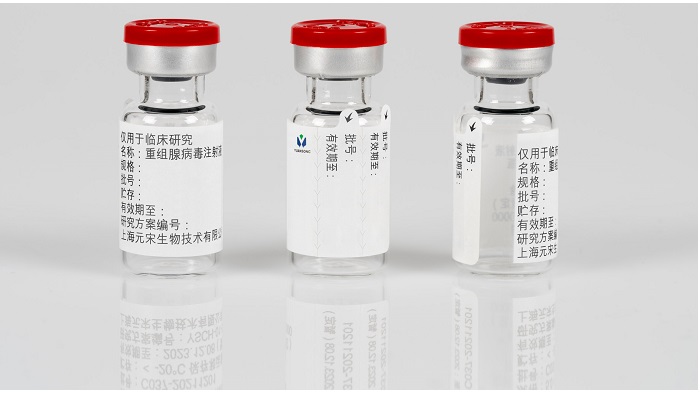
Virotherapy is an immunotherapy in which patients receive injections with oncolytic viruses. The treatment employs viruses to infect tumor cells, selectively destroying malignant cells. At Yuan Song Biotechnology, the active pharmaceutical ingredient (API) for its vial-filled virotherapy cancer treatment is especially sensitive, posing labeling challenges. The sensitive API must be stored and transported at low temperatures, and is subsequently thawed into a liquid before being administered.
These temperature differences place high demands on the reliable adhesion of the marking and documentation label attached to unit-dose vials. In addition, prior to administering the drug, healthcare staff must check if the medication is flawless and the liquid is clear. A transparent inspection window must be integrated into the label for that purpose. The label also must provide adequate space for important information, and contain two removable parts for documenting the administration of the medication in the patient and hospital files for the clinical trial.
For Yuan Song Biotechnology, the temperature stability and multifunctional benefits of Schreiner MediPharm’s customized Pharma-Comb IL Label enabled reliable and clear product marking, from storage to transportation to dispensing. The two removable documentation labels have tabs allowing easy and efficient peel-off and attachment to patient files. Thanks to the transparent inspection window, the vial liquid can be reliably checked prior to being administered. Thus, the Pharma-Comb IL Label from Schreiner MediPharm helps optimize clinical trial processes and supports patient safety.