The protection of patient health starts with ensuring the safety of pharmaceutical products even before they reach healthcare providers. Every recall caused by defective or leaking containers is one too many. As the pharmaceutical industry continues to evolve towards safer processes, a key trend is driving positive change: the growing demand for leak detection complementary to visual inspection.
Visual inspection has been the industry standard for decades and remains indispensable. It helps to detect unwanted particles and cosmetic defects before the drugs reach the patients. However, the risk of tiny holes, invisible to the human eye and high-resolution cameras, might remain. They enable microorganisms or other contaminants to enter the primary packaging.
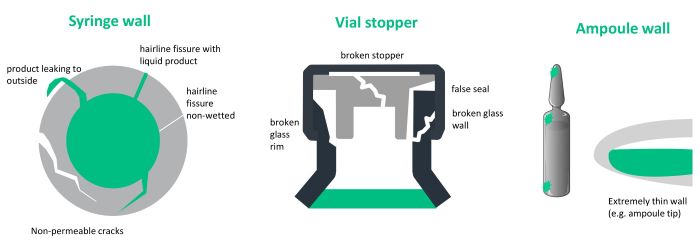 Container closure integrity testing (CCIT) technologies check each individual container for the smallest leaks. They complement visual inspection and ensure the integrity of the containers. Considering the latest advancements in drug development in both sensitive and expensive therapeutic areas such as immunotherapy, gene therapy, oncology, and biologics, 100% integrity testing is of particular interest.
Container closure integrity testing (CCIT) technologies check each individual container for the smallest leaks. They complement visual inspection and ensure the integrity of the containers. Considering the latest advancements in drug development in both sensitive and expensive therapeutic areas such as immunotherapy, gene therapy, oncology, and biologics, 100% integrity testing is of particular interest.
The role of Annex 1
 Comparing the 2018 version of EU GMP Annex 1 with the latest version (effective since August 2023), container closure inspection now plays a much more prominent role, both in scope and detail of the guideline. For the first time, Annex 1 is harmonized with the WHO and 56 PIC/S member countries, significantly increasing its global relevance and impact. For example, Chapter 8.22 emphasizes the importance of CCIT to ensure the safety and quality of pharmaceutical products. The guideline requires 100% integrity testing for fusion-sealed containers (up to 100 ml). One of the methods adaptable for commercial use is high-voltage leak detection (HVLD).
Comparing the 2018 version of EU GMP Annex 1 with the latest version (effective since August 2023), container closure inspection now plays a much more prominent role, both in scope and detail of the guideline. For the first time, Annex 1 is harmonized with the WHO and 56 PIC/S member countries, significantly increasing its global relevance and impact. For example, Chapter 8.22 emphasizes the importance of CCIT to ensure the safety and quality of pharmaceutical products. The guideline requires 100% integrity testing for fusion-sealed containers (up to 100 ml). One of the methods adaptable for commercial use is high-voltage leak detection (HVLD).
These regulatory changes reflect a global market trend towards stricter safety standards, as well as the growing adoption of HVLD as a standard requirement in automated inspection systems. Chapter 8.22 further mentions that visual inspection alone is insufficient to check container integrity, making the combination with other testing methods like HVLD even more important. Chapter 8.23 clarifies that products using systems other than fusion should also be tested for integrity using validated methods. HVLD is specifically recommended as a validated method in both Annex 1 and in chapter 1207 of the United States Pharmacopeia (USP), where a whole chapter is dedicated to methods for assessing package integrity.
Understanding leak formation
 Leaks can occur at various stages of the container lifecycle, making leak testing a crucial part of final quality inspection. Tiny leaks can already develop during the glass formation process due to imperfections or stress in the glass. If containers are overfilled or the filling equipment is not properly calibrated, overpressure and potential breaches might occur during the actual filling process. Furthermore, transport-related issues such as rough handling, high-speed contact with other containers, or temperature changes can cause cracks or fractures in the glass. Apart from process-related leaks, vulnerable container parts such as the tip of an ampoule are susceptible to damage during handling and storage.
Leaks can occur at various stages of the container lifecycle, making leak testing a crucial part of final quality inspection. Tiny leaks can already develop during the glass formation process due to imperfections or stress in the glass. If containers are overfilled or the filling equipment is not properly calibrated, overpressure and potential breaches might occur during the actual filling process. Furthermore, transport-related issues such as rough handling, high-speed contact with other containers, or temperature changes can cause cracks or fractures in the glass. Apart from process-related leaks, vulnerable container parts such as the tip of an ampoule are susceptible to damage during handling and storage.
Leaks can be permeable and non-permeable, depending on the nature of the breach and the container’s design. Permeable leaks involve breaches that permit the passage of liquids. They result in a loss of product content, leading to potential contamination. Non-permeable leaks, on the other hand, refer to breaches that allow the passage of gases or vapors through the container wall without a significant flow of liquid. They can also weaken the container’s material. Over time, the container becomes more prone to breakage or failure, which can lead to leaks while processing.
How HVLD ensures leak-free containers
HVLD is a precise, non-destructive method used to assess the integrity of pharmaceutical containers. For the process to work effectively, the liquid inside the container must be conductive. If the container is intact, only a minimal amount of high voltage current can flow through it. If, however, the container has a small crack or hole, the conductive liquid allows a large current to pass through, triggering the system to detect the leak. The container is then automatically rejected.
 The effectiveness of HVLD is influenced by several key factors that must be carefully controlled when setting up the inspection process. For example, higher conductivity in the product allows for easier detection; thicker glass containers require higher voltage or longer inspection times; ambient temperature, humidity, as well as ozone levels can all affect detection, potentially causing false positives or rejects. Moreover, fine control of the high voltage is key. While high voltage set to a low value might miss small leaks, too high values could trigger false rejects. The correct positioning and alignment of the HVLD electrodes in contact with the containers is also crucial for reliable results.
The effectiveness of HVLD is influenced by several key factors that must be carefully controlled when setting up the inspection process. For example, higher conductivity in the product allows for easier detection; thicker glass containers require higher voltage or longer inspection times; ambient temperature, humidity, as well as ozone levels can all affect detection, potentially causing false positives or rejects. Moreover, fine control of the high voltage is key. While high voltage set to a low value might miss small leaks, too high values could trigger false rejects. The correct positioning and alignment of the HVLD electrodes in contact with the containers is also crucial for reliable results.
Expertise and training are the key
Implementing HVLD technology requires experts who ensure these factors are optimized. This calls for thorough training for operators and maintenance staff to deliver the safest and most reliable leak detection. Apart from training, histograms and batch reports can offer valuable insights and help to collect experiences in key measurements and critical parameters. These tools are essential both for monitoring performance and when developing further recipes, as they highlight the performance of current settings based on every unique product and its production environment.
Finally, a rigorous validation of the system makes sure that the inspection process functions correctly and meets safety standards. It involves calibrating the system with test containers, which have known leaks and setting the appropriate sensitivity and high voltage. Containers used for validation can either be self-made crack samples or samples with laser-drilled holes. Control samples are used to confirm accuracy, and repeatability tests are conducted to check the system’s reliability across different containers and operators.
Benefits of integrating HVLD with AVI
HVLD supplements visual inspection by detecting leaks that neither cameras in Automated Visual Inspection (AVI) systems nor the human eye in manual inspection can. In the past, both AVI and HVLD inspection were performed with stand-alone machines that required a larger footprint, higher investments, and the connection to either upstream processes or additional transportation. By integrating HVLD into AVI, pharmaceutical manufacturers receive a seamless process in one single system that operates at the same speed as the AVI machine. Although the initial investment in HVLD equipment can be a barrier for smaller manufacturers, reduced recalls, less rework, and faster production times make it a worthwhile choice and are driving its increased demand.
HVLD is an effective and validated method to detect leaks in pharmaceutical containers. It aligns seamlessly with the updated EU GMP Annex 1 guidelines, which focus on contamination control and sterility in pharmaceutical manufacturing. By providing sensitive and real-time detection of leaks, HVLD plays a crucial role in ensuring safety, sterility, and integrity. Safety standards in the industry are evolving, and visual inspection is no longer sufficient for many pharmaceutical manufacturers. The combination of AVI and HVLD is increasingly becoming a requirement for the investment in new inspection machines and contributes significantly to improving quality standards and ensuring patient safety.






















