Key Takeaways
- 80% of clinical trials experience enrolment delays and massive sunk costs with underperforming sites.
- Only 4% of US physicians participate in clinical research, leaving 96% of patients inaccessible under other HCPs’ care.
- Tokenization links Citeline’s proprietary data; specifically, 300+ million claims lives, 245+ million lab lives & 55 million+ EMR patient lives with 1.7+ million HCP national provider identifiers (NPIs).
- Direct-to-patient traditional advertising recruitment campaigns fail with complex protocol criteria, most commonly in oncology and rare disease protocols.
- Last-mile operationalization requires fair market value compensation and patient concierge services.
By Matt Holms, Vice President, Commercial Patient Engagement & Recruitment
Clinical trial site selection remains one of the pharma industry’s most expensive guessing games. The cost to set up a site can range from anywhere from $40,000 to $60,000.
Yet only about 4% of physicians in the United States participate in clinical research. The chart below outlines the significant headwinds facing sponsors and sites for patient recruitment in the clinical trial industry.
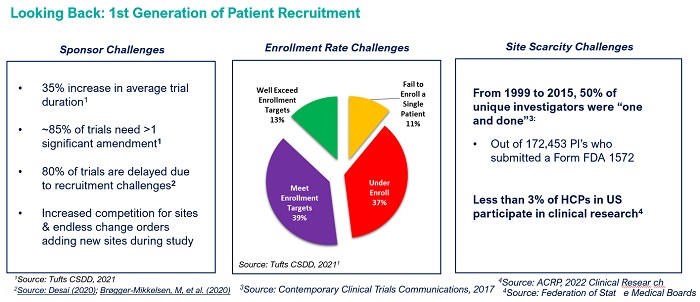 Traditional patient recruitment approaches can’t solve a scarcity problem
Traditional patient recruitment approaches can’t solve a scarcity problem
The first generation of patient recruitment was the assumption that the clinical research sites that sponsors selected would enroll 100% of the trial with patients from their own database alone. Sponsors invest substantial resources into site selection, partnering with CROs that claim superior investigator networks, that all too often face rescue scenarios into Phase II and Phase III studies.
 The second generation of patient recruitment approach attempted to circumvent site database limitations through direct-to-patient advertising campaigns often using a variety of outreach tactics to target patients not known to the study sites. While traditional patient recruitment campaigns have been effective for chronic indications, complex protocol designs expose this approach’s fundamental weakness.
The second generation of patient recruitment approach attempted to circumvent site database limitations through direct-to-patient advertising campaigns often using a variety of outreach tactics to target patients not known to the study sites. While traditional patient recruitment campaigns have been effective for chronic indications, complex protocol designs expose this approach’s fundamental weakness.
Many research sites are understaffed, and this is where the process often falls down. Rescue campaigns are put in place as a last-ditch effort, but if the protocol is complex, patients are not able to self-report answers to critical I/E criteria in the initial prescreener. This leads to high volumes of referrals that are not qualified against the key I/E criteria, overwhelming sites and further increasing burden.
 Tokenization as infrastructure: Linking RWD & proprietary data for visibility at the patient, disease, and provider Level
Tokenization as infrastructure: Linking RWD & proprietary data for visibility at the patient, disease, and provider Level
Citeline’s vision with the third generation of patient recruitment replaces assumption-based planning with data-linked patient identification at population scale. This involves aggregating both proprietary and real-world data across multiple dimensions, creating visibility into patient populations that traditional site databases cannot access.
Tokenization technology aggregates disparate data sources and eliminates silos by assigning unique patient tokens that link patient longitudinal data from sources such as EMR, lab, and claims information into unified patient profiles. These patient tokens can be linked to National Provider Identifier (NPI) numbers for HCPs, as well as facility NPIs, creating visibility into who treated patients, when they were treated, how they were treated, where treatment occurred, and what therapies were administered.
The IRT data redaction implications to results-based pricing
Privacy protection measures created unintended operational consequences that have destabilized recruitment vendor accountability models. Historically, recruitment companies verified that prescreened referred patients either consented or randomized by matching Interactive Response Technology (IRT) data using unique identifiers like first and last initials combined with full date of birth. Sites updated vendor portals inconsistently, making IRT data the gold standard reconciliation mechanism for results-based pricing contracts for recruitment vendors meeting patient delivery milestones.
There has been a big shift in the industry to redact that data where sponsors’ IRT systems now typically only collect year of birth and/or gender now. In order to corroborate that they actually delivered a consented or randomized patient — as this is the only mechanism for remuneration — many vendors have resorted to bombarding sites with communications to validate if referrals signed an informed consent form (ICF) and ultimately randomized.
The accountability dilemma extends to referring physicians who operate outside investigator networks too. Non-investigator physicians face three simultaneous barriers: time required for chart review and coordination, revenue loss from reimbursement structures, and patient loss from clinical continuity. Revenue and continuity concerns, such as violating anti-kickback statutes, require broader solutions to facilitate patient access to clinical trials.
The Last Mile: Why data identification is only a part of the solution
Citeline launched its tokenized patient match recruitment solution nearly two years ago, generating critical learnings about the gap between patient identification and patient randomization. The biggest learning relates to services and support infrastructure that facilitates getting a patient to the site, which the industry has recently coined “the Last Mile.”
Cross-vendor collaboration defines our strategic approach. The company partners with organizations that have also tokenized data through Datavant, enabling token-sharing for direct-to-patient outreach where appropriate. Other partnerships focus on providing “last mile” patient concierge services that can operate on the protected health information (PHI) side to follow up with identified patients
Measurement challenges persist where tracking conversion from identification to randomization requires coordination across MSLs, contracting teams, patient services, and sites — a cross-functional orchestration that extends beyond traditional recruitment vendor scope.
From proprietary advantage to collaborative ecosystem
Third-generation recruitment represents a paradigm shift from simply hiring the best sites to identifying patients both in and around sites at a specific moment in time based on their clinical pathway. It is important to frame patient access as both a data and service delivery challenge rather than simply a site selection problem.
Sponsors increasingly use tokenized RWD for protocol design and feasibility analysis before finalizing inclusion-exclusion criteria, preventing enrollment challenges through evidence-based protocol optimization. This trend still has a long way to go, with significant upside for the industry.
Therapeutic area segmentation matters: Direct-to-patient campaigns retain value in chronic disease indications where patients can appropriately answer prescreening questions, while oncology, rare disease and any complex indications’ protocols require data-driven identification of protocol-matched patients and their treating physicians.
Third-generation recruitment will succeed not through just proprietary advantage but through orchestrated collaboration that combines RWD/proprietary data, tokenization, artificial intelligence (AI), and complex services collaboration into randomized patients.






















